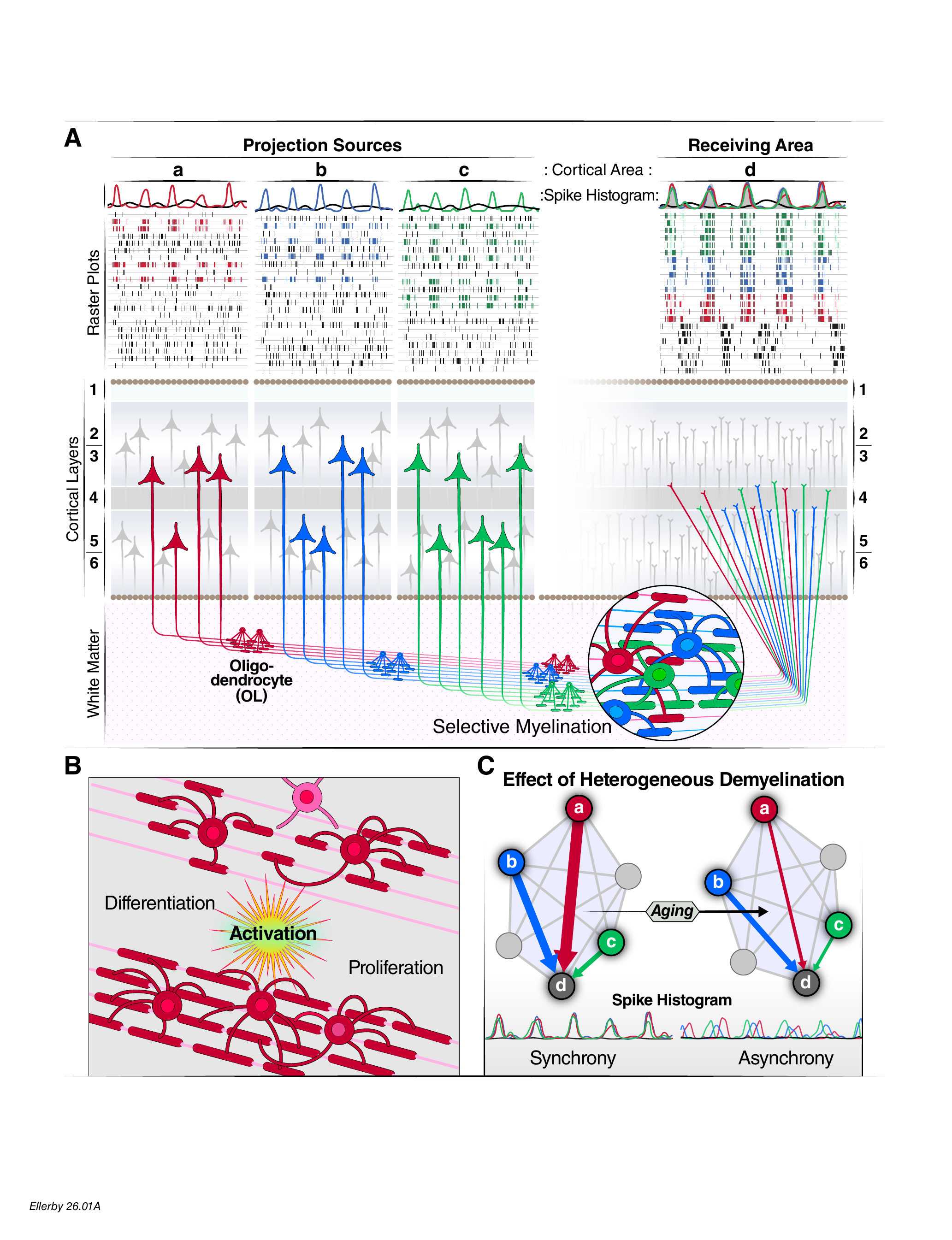
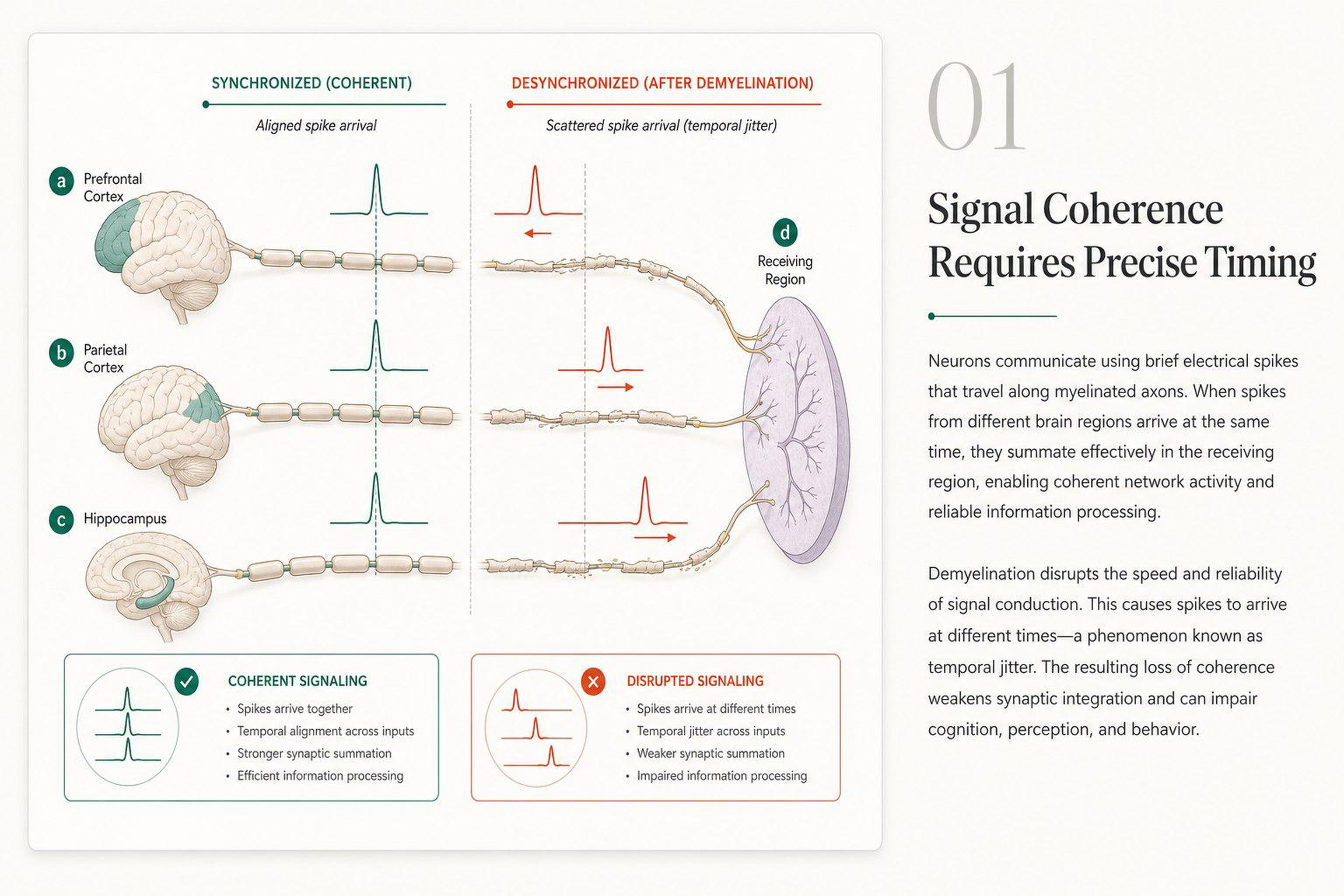
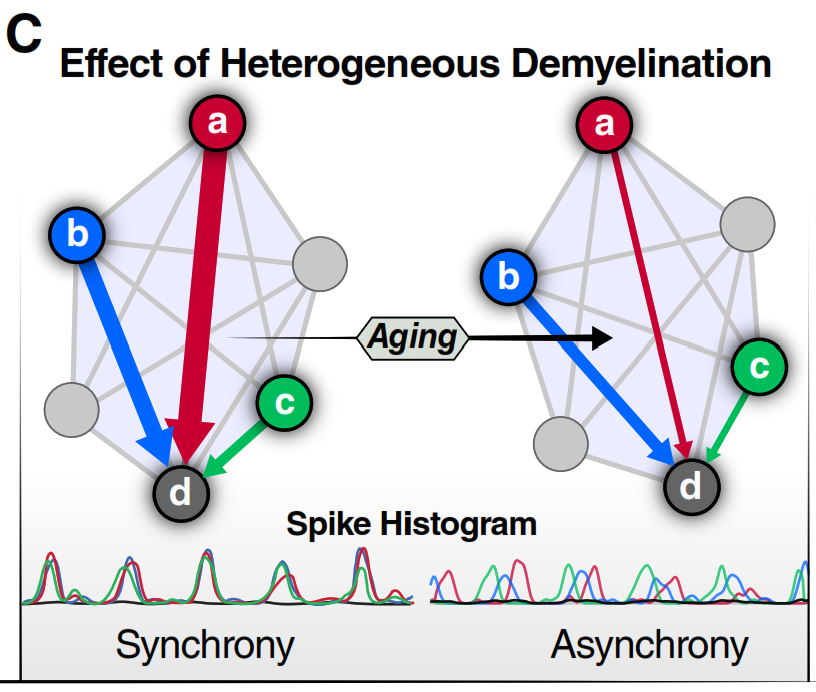
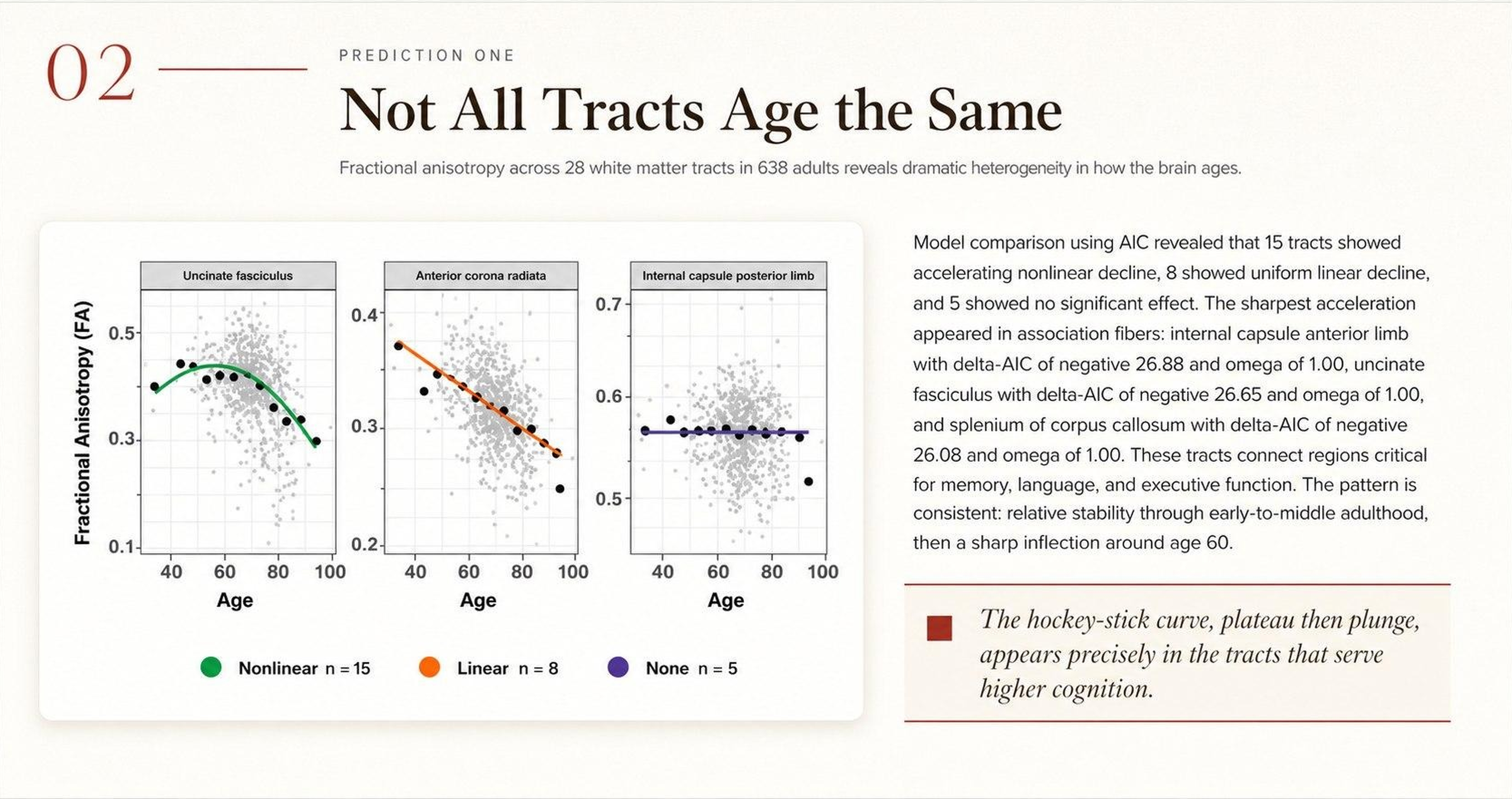
The brain is a network of roughly 86 billion neurons organized into dozens of specialized regions, each handling a different job — recognizing faces, planning tomorrow, reading this sentence. Thinking, remembering, and deciding require many of these regions to activate at the same time, in precise coordination.
The concert hall. Imagine an orchestra whose musicians sit in different buildings across a city, connected only by cables carrying their sound. For the symphony to work, every note must arrive at the mixing board within milliseconds of the right beat. If even one cable introduces a delay, that section falls out of rhythm and the whole piece suffers.
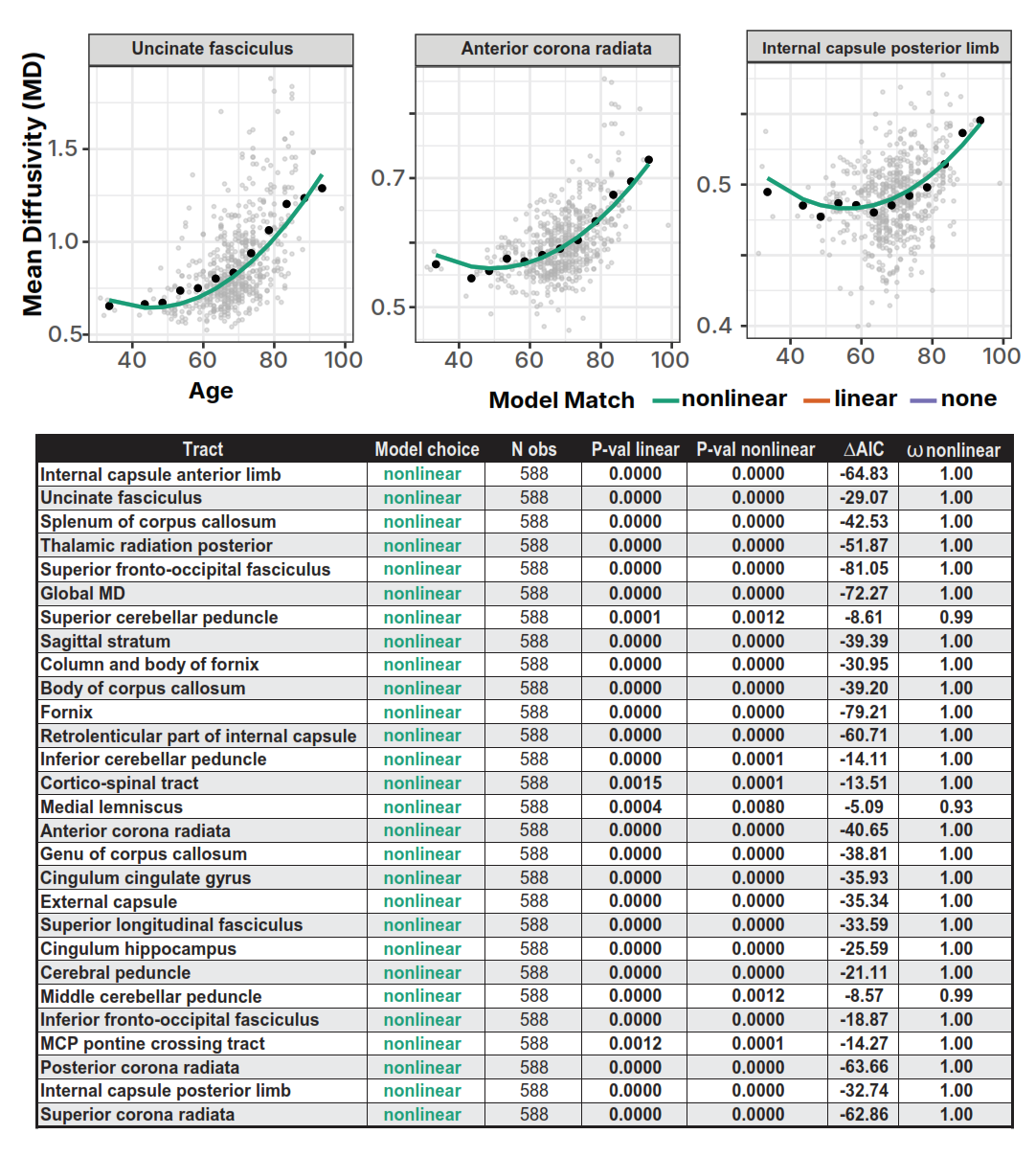
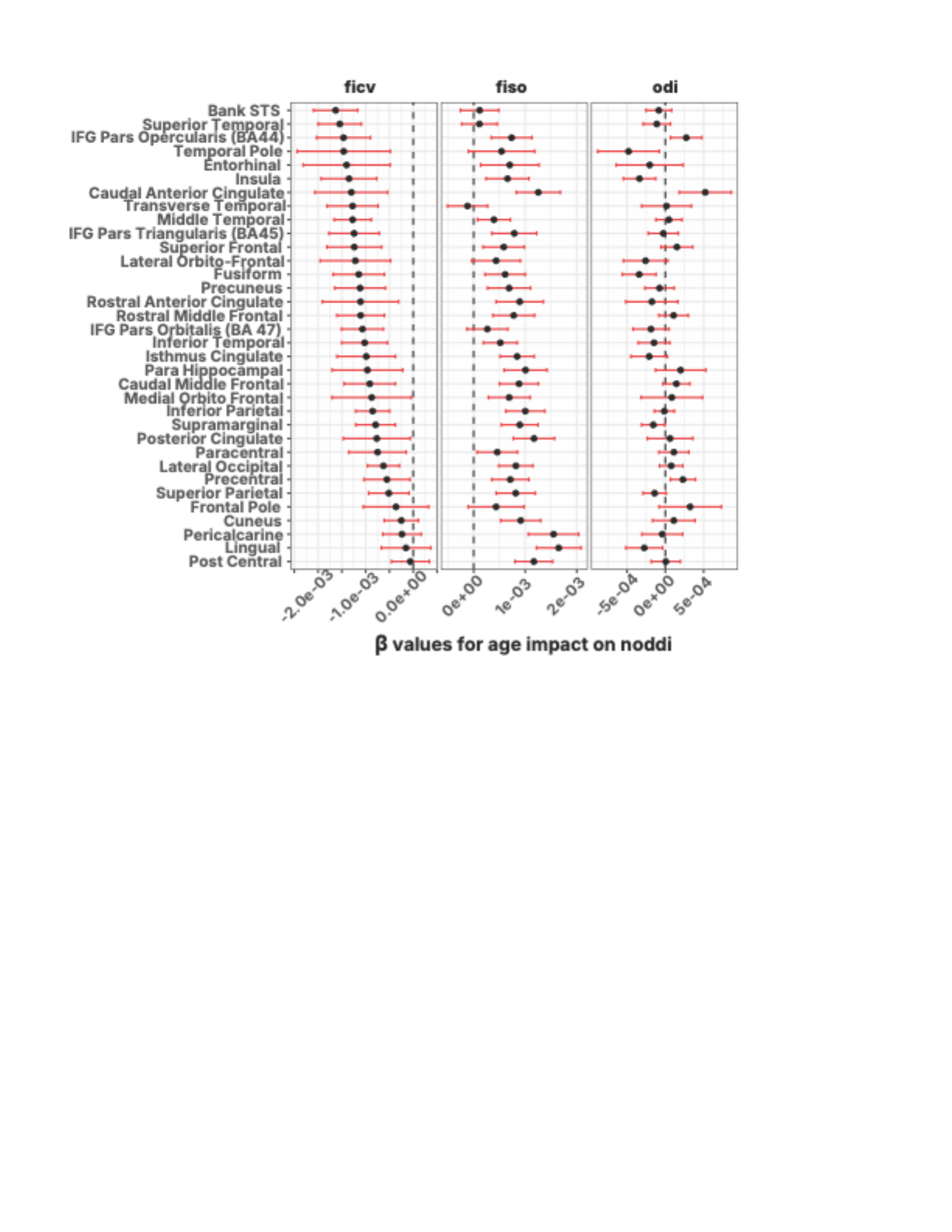
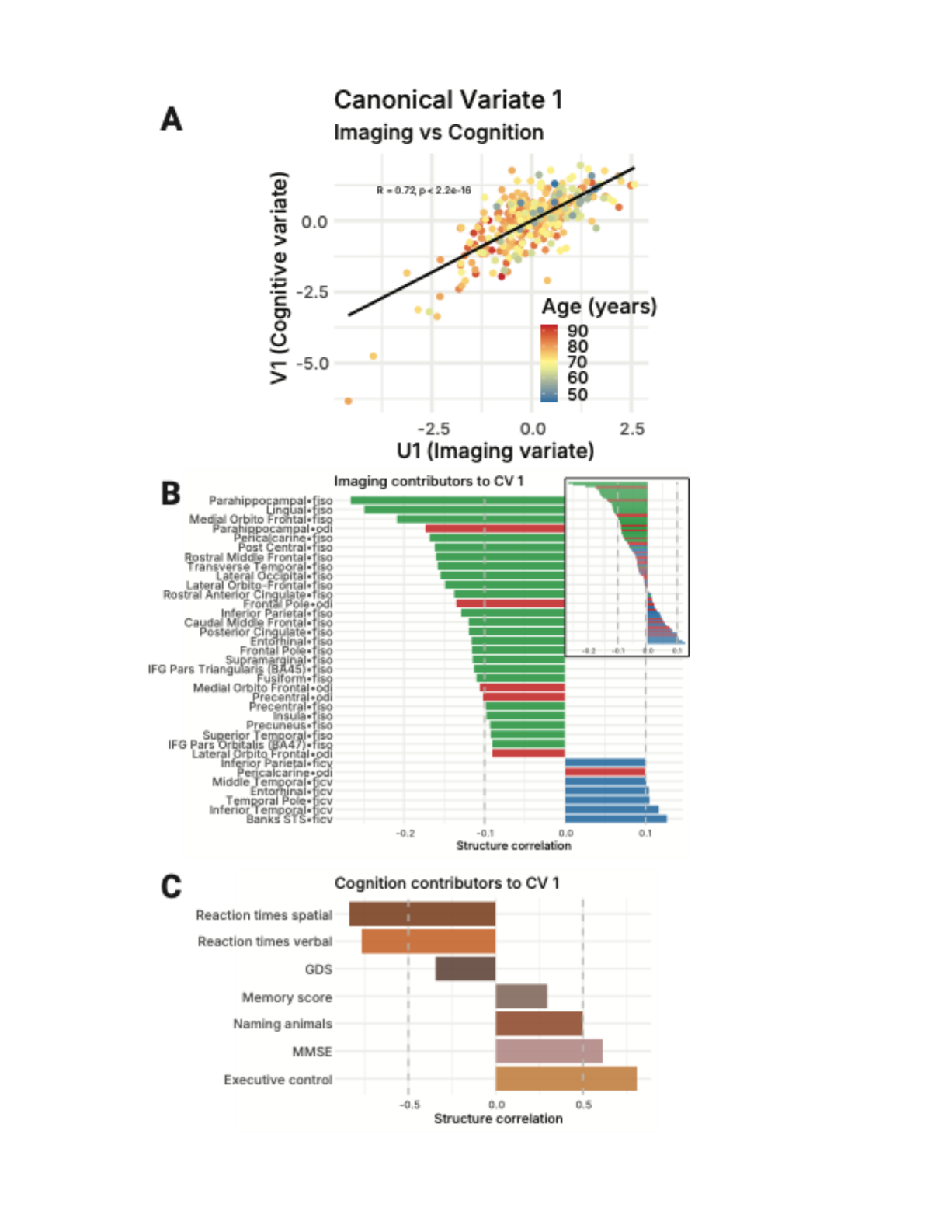
In the brain, those cables are white matter tracts: bundles of long nerve fibers that link distant regions. Each fiber is wrapped in a fatty coating called myelin. Myelin does two things: it speeds up electrical signals (from ~1 m/s to ~100 m/s) and, critically, it keeps their timing precise.